Description
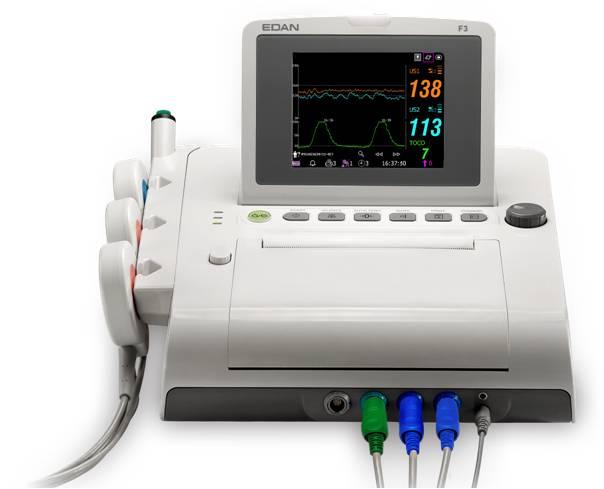
EDAN F3 CTG Monitor
Fetal Monitor F3
Colour TFT Screen
Easy to Carry and Operate
Advanced FHR detection technology
Built in Printer
Battery
Data management on PC
Suitable for Outpatient Service
- Foldable color screen Foldable color screen
- Backup memory for seamless monitoring Backup memory for seamless monitoring
- Built-in long life thermal printer Built-in long life thermal printer
- Compatible with 150/152 mm thermal recording paper Compatible with 150/152 mm thermal recording paper
- Large numeric and graphic display for ease of readability Large numeric and graphic display for ease of readability
- FHR signal quality indicator FHR signal quality indicator
- Signals Overlap Verification to separate twins FHR Signals Overlap Verification to separate twins FHR
- Fast printing of stored traces Fast printing of stored traces
- Long life Li-ion battery guarantees the mobility of F3 Long life Li-ion battery guarantees the mobility of F3
- PC management software and central monitoring system supportive PC management software and central monitoring system supportive
- Convenient probe rack and compact design Convenient probe rack and compact design
- USB port enables memory expansion USB port enables memory expansion
- DECG & IUP parameters optional
- DECG & IUP parameters optional
EDAN F3 CTG Monitor Product Specifications
Environmental Specifications
The monitor may not meet the performance specifications given here if stored or used outside the specified temperature and humidity ranges.
Working
Temperature: +5 ºC ~ + 40 ºC ( +41 ºF ~ +104 ºF)
Relative Humidity: 15% ~93% (non-condensing)
Atmospheric Pressure: 86 kPa ~ 106 kPa
Transport and Storage
Temperature: -20 ºC ~ +55 ºC (-4ºF ~ +131 ºF)
Relative Humidity: 15% ~ 93% (non-condensing)
Atmospheric Pressure: 70 kPa ~ 106 kPa
Physical Specifications
Dimensions and Weight:
Size (depth x width x height) : 350mm x 300mm x 104mm
Weight: Approx. 3.5 kg
Power Supply
Operating Voltage: 100V-240V~
Operating Frequency: 50Hz/60Hz
Input Power : 70VA
Battery: 14.8VDC/5000mAh
Standards Compliance
IEC 60601-1:2005/A1:2012, EN 60601-1:2006/A1:2013,
IEC 60601-1-2:2014, EN 60601-1-2:2015, IEC/EN 60601-2-37
Anti-electric Shock Type: Class I equipment with internal power supply
Anti-electric Shock Degree:
FHR1, FHR2, TOCO, FM, FS: BF
IUP: BF
DECG: CF
Degree of Protection against Harmful Ingress of Water for the Transducers:
IPX 8 (FHR1,FHR2,TOCO)
EMC: CISPR 11 Group 1 Class A
Working System: Continuous running equipment
Earth Leakage Current (Limit):
N.C.: 500μA
S.F.C.: 1000μA
Enclosure Leakage Current (Limit)
N.C.: 100μA
S.F.C.: 500μA
Patient Leakage Current (Limit)
N.C. d.c.: 10μA 50μA
S.F.C. a.c. 10μA 50μA
Patient Auxiliary Current (Limit)
N.C.: d.c. 10μA 50μA
S.F.C.: a.c. 10μA 50μA
Display
LCD Size: 5.6”|
Resolution: 640 (RGB) x 480
Signal Interface DB9, RJ45 network interface
Ultrasound Transducer
Weight: 190g
Cable Length: 2.5m
Dimension: 88mmÅ~35mm
TOCO Transducer
Weight: 180g
Cable Length: 2.5m
Dimension: 88mmÅ~35mm
Remote Event Marker
Length: 2.5m
Weight: 56g
Performance Specifications
Ultrasound
Technique: Ultrasound Pulse Doppler with autocorrelation
Pulse Repetition Rate: 2 KHz
Pulse Duration: 92 μs
Ultrasound Frequency: ( 1 .0 ±10%) MHz
p- <1 MPa
Iob <10 mW/cm2
Ispta <100 mW/cm2
*Isata <20 mW/cm2
*Isppa.3<190W/cm2
*Ispta.3<94mW/cm2
|Max Output Power <15mW
Effective Radiating Area: 628 mm2 ± 15%
*FHR Measurement Range: 50 bpm ~ 240 bpm
*Resolution: 1 bpm
*Accuracy: ±2 bpm
*Temperature Rise:
When applied to the patient, the ultrasound transducer may warm slightly (less than 2°C (3.6ºF) above ambient temperature. When NOT applied, at the ambient temperature of 40°C (104ºF), the ultrasound transducer may reach the highest temperature of 43°C (109.4ºF).
Dielectric Strength: > 4000Vrms
ISATA@ the transducer face: 1.865 mW/cm2
Entrance beam: 6.08 cm2
Measurement uncertainties for ISATA: +26.6%
Measurement uncertainties for ultrasonic power: +26.6%
DECG (optional)
Technique: Peak-peak detection technique
*DFHR Measurement Range: 30bpm ~ 240bpm
*Resolution: 1bpm
*Accuracy: ±1bpm
Input Impedance: > 10M (Differential, DC50/60Hz)
Input Impedance: > 20M (Common Mode)
CMRR: > 110dB
Noise: < 4μVp
Skin Voltage Tolerance: ±500mV
Fetal Input Voltage Current: 20μVp ~ 3mVp
*TOCO Range: 0 ~ 100
*Non-linear Error: ±10%
TOCO
Baseline Drift due to
Temperature Changes
≤ 1 unit/min/°C (free air)
≤ 5 units/min/°C (underwater)
*Resolution: 1
Zero Mode: Automatic/ Manual
IUP (optional)
*Pressure Range: 0mmHg ~100mmHg (0.0 kPa~13.3 kPa)
Sensitivity: 5μV/V/mmHg
*Non-linear Error: ±3mmHg (±0.4 kPa)
*Resolution: 1mmHg (0.1 kPa)
Zero Mode: Manual
AFM
Technique: Pulsed Doppler ultrasound
Marking Manual fetal movement mark
NOTE:The essential performance is marked with an asterisk *.
Recorder Specifications
Paper:
Z-fold, thermosensitive (compatible with GE and PHILIPS recorder paper)
Paper width: 152mm (GE), 150mm (PHILIPS)
Effective printing width:
110mm (American Standard)
120mm (International Standard)
FHR printout width:
|70mm (American Standard)
80mm (International Standard)
FHR scaling:
30bpm/cm (American Standard)
20bpm/cm (International Standard)
TOCO printout width: 40mm
TOCO scaling: 25%/cm
Printing speed:
Standard Speed (Real-Time Traces ): 1 cm/min, 2 cm/min, 3 cm/min
Fast Print Speed (Stored Traces): Up to 15mm/sec
Accuracy of data: ± 5% (X axis)
Accuracy of data: ± 1% (Y axis)
Resolution: 8 dots/mm
Record Information:
FHR1 trace/mark, FHR2 trace/mark, TOCO trace, AFM trace/black mark, fetal movement mark, event mark, fetal stimulation mark, AUTO-zero symbol, date, time, printing speed, ID, name, FHR2 Offset etc.
Lithium-ion Battery Specifications
Battery Specification:
01.21.064143 Nominal Capacity 5000mAh
Working Time: >7 hours (depending on configuration)
Nominal Voltage 14.8V
Charge Time <6 hours
Cycle Life ≥300 times
Additional information
| Weight | 10 kg |
|---|